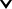Graphite
What is graphite
Graphite is an industrial mineral with unique physical properties which includes superior thermal/electrical conductivity and generally occurs in one of three forms – Microcrystalline or amorphous, Crystalline lump or vein and Crystalline flake.
An industrial mineral has many unique physical properties:
- Superior thermal/electrical conductivity
- Stable wide temperature range
- Excellent lubrication
- Malleable
- Resistant to chemical attack
- Fire retardant and thermally efficient building products